IV Set Varieties: An Overview of Available Intravenous Set Types
Introduction
In modern clinical practice, iv set is a topic every healthcare professional must understand thoroughly. Proper knowledge of this subject directly impacts patient safety and treatment effectiveness across all clinical settings.
Core Clinical Concepts
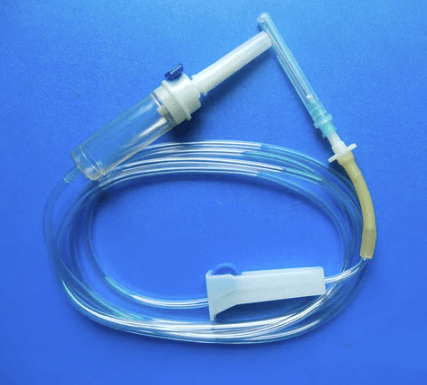
An intravenous (IV) set consists of several interconnected components that work together to deliver fluids, medications, or blood products directly into a patient’s bloodstream. Understanding these parts helps clinicians troubleshoot issues and maintain patient safety.
The Spike and Drip Chamber
The spike is the sharp entry point that pierces the IV bag or bottle, connecting directly to the drip chamber. The drip chamber allows clinicians to visually monitor fluid flow rate. Proper insertion of the spike is critical to maintaining a sterile environment throughout the infusion process.
Tubing and Flow Regulator
The flexible PVC tubing carries fluid from the bag to the patient. Modern IV sets include a roller clamp that allows precise control over the drip rate. Tubing length typically ranges from 150 cm to 200 cm depending on the clinical application and patient positioning requirements.
Practical Applications in Healthcare
For nurses and physicians, familiarity with every component is not just academic. It directly impacts patient outcomes. A malfunctioning drip chamber or kinked tubing can interrupt therapy and cause serious complications including fluid imbalance or medication errors.
- Always verify sterility and expiration date before use
- Use aseptic technique during all connections and disconnections
- Document the time of setup and rate of infusion in patient records
- Monitor the infusion site regularly for signs of infiltration or phlebitis
- Change equipment per institutional protocol to prevent infection
Safety and Quality Considerations
Before connecting an IV set, every component must be visually inspected. Check packaging integrity, look for cracks or discoloration in tubing, and verify the expiration date. The drip chamber should be free of bubbles, and the filter should be intact and undamaged.
Frequently Asked Questions
FAQ 1: What should I do if I notice a defect in IV equipment?
Any defective IV equipment must be removed from service immediately. Document the defect, report to your supervisor, complete an incident or adverse event report, and retain the defective product for investigation if required by institutional policy.
FAQ 2: How does patient age affect equipment selection?
Patient age significantly influences equipment selection. Neonates and infants require micro-drip sets and smaller cannulas. Pediatric patients need smaller volumes with more precise control. Adults typically use standard macro-drip sets for routine fluid therapy.
See also: Wearable Technology Innovations in Healthcare
FAQ 3: Are there environmental considerations for disposing of IV equipment?
Used IV equipment including tubing, needles, and filters must be disposed of as clinical waste following local regulations. Sharp components go into approved sharps containers. Tubing and non-sharp parts go into clinical waste bins.
Conclusion
Thorough knowledge of iv set is an essential component of professional clinical practice. By understanding the details covered in this guide, healthcare providers are better prepared to deliver safe, effective, and evidence-based care to every patient.